New C. diff Antibiotics: CRS3123 and Ibezapolstat Show Remarkable Phase 2 Results
Other Categories
For the millions of Americans who have lived through a C. diff infection or lost a loved one to one, the promise of better treatments is not an abstract hope. It is deeply personal. This month, two investigational drugs are generating significant scientific interest, and we want to make sure patients and survivors have the information it needs to understand what these developments could mean.
The core problem with existing treatments like vancomycin is that while they work reasonably well at curing an initial infection, they are broad-spectrum antibiotics. They disrupt the very gut microbiome that serves as one of the body’s primary defenses against C. diff. Once that ecosystem is destabilized, recurrence becomes far more likely. Two new drugs in clinical development take direct aim at this problem with different approaches — and the results so far are encouraging.
CRS3123: A Narrow-Spectrum Antibiotic That Spares Your Gut
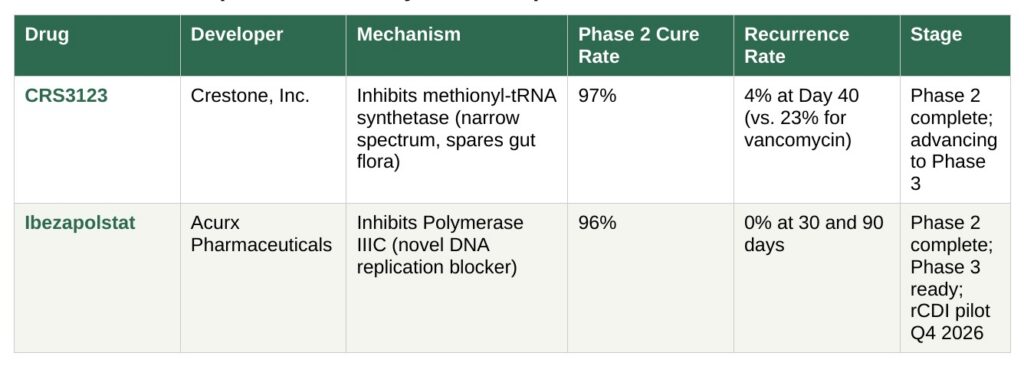
Developed by Boulder, Colorado-based biopharmaceutical company Crestone, CRS3123 is a small-molecule antibiotic with a precise, narrow target: a specific bacterial enzyme called methionyl-tRNA synthetase. This enzyme is critical to C. diff’s survival but is absent in the bacteria that make up the healthy human gut microbiome. CRS3123 can attack the pathogen while leaving the surrounding community of beneficial microbes largely intact, exactly the kind of treatment the C. diff field has long needed.
The Phase 2 clinical trial, recently published in The Lancet Infectious Diseases, enrolled 58 adults with a primary or first-recurrence episode of C. diff across 14 sites in the U.S. and Canada. Participants received one of two doses of CRS3123 (200 mg or 400 mg twice daily) or the standard comparator, vancomycin, for 10 days.
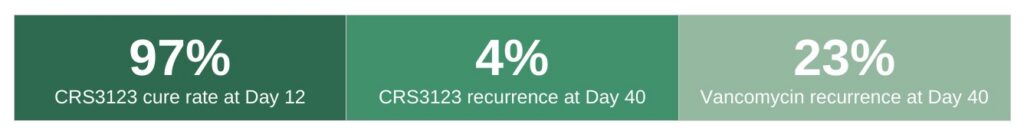
The cure rates were comparable between CRS3123 and vancomycin, 97% for patients receiving either dose of CRS3123, compared to 93% for vancomycin at Day 12. But the headline finding is in the recurrence data. Only 4% of CRS3123 patients experienced C. diff recurrence at Day 40, compared to 23% in the vancomycin group. Among patients taking the 200 mg dose specifically, there were zero recurrences through Day 40.
Equally important, the drug was well tolerated. CRS3123 showed no serious treatment-emergent adverse events. Side effects that did occur were mild to moderate and primarily gastrointestinal.
“Treatment of C. difficile infection remains in urgent need of agents that spare normal gut microbes, so they can reconstitute the microbiome and prevent further recurrences of CDI. The findings of this study support CRS3123 as such a candidate for further development.”
— Dr. Thomas Louie, MD, University of Calgary, Principal Investigator
In microbiome analyses from the trial, CRS3123 better preserved gut microbiome diversity compared to vancomycin — for both alpha and beta diversity — suggesting a healthier gut ecosystem that can fend off future C. diff colonization.
The FDA has already granted CRS3123 both Qualified Infectious Disease Product (QIDP) and Fast Track designations. The National Institute of Allergy and Infectious Diseases (NIAID) has committed $4.5 million in new funding to support further microbiome studies and the optimization of manufacturing processes, advancing the path toward Phase 3 trials.
Ibezapolstat: A Novel Mechanism With a 96% Cure Rate and No Recurrences
The second drug generating excitement in the C. diff community comes from Acurx Pharmaceuticals. Ibezapolstat works through an entirely different mechanism than any existing C. diff treatment. It targets Polymerase IIIC, the main enzyme responsible for replicating bacterial DNA in many Gram-positive bacteria, including C. diff and Staphylococcus aureus. Because this target is distinct from those of current antibiotics, ibezapolstat is unaffected by resistance to existing drug classes — a meaningful advantage given the broader global crisis of antimicrobial resistance.
The Phase 2 results for ibezapolstat were striking. The drug demonstrated a 96% clinical cure rate for C. diff infection, with no recurrences observed at either 30-day or 90-day follow-up. That is not a typo: zero recurrences in the follow-up window. While the trial was relatively small and further study is needed, these early results are the kind of signal that gets researchers and patient advocates paying close attention.

Acurx has already received final guidance from both the FDA and the European Medicines Agency (EMA) for ibezapolstat’s pivotal Phase 3 trials. The company has also announced plans for a new trial specifically targeting patients with multiple recurrent C. diff — an open-label pilot study enrolling 20 patients who have experienced at least three C. diff episodes in the past 12 months.
Trial start-up activities are expected to initiate later this month, with the first patient enrollment anticipated in the fourth quarter of 2026.
Why This Matters for the C. diff Community
For patients and families who have been through recurrent C. diff, these are significant developments. The current standard of care cures many patients on the first try. But for those who experience recurrence, each subsequent episode becomes harder to treat, and the emotional and physical toll compounds. Both CRS3123 and ibezapolstat could address this challenge.
Important Caveats: Promising, But Not Yet Available
It’s important to note that neither of these drugs is approved or currently available outside of clinical trials. Phase 2 studies involve relatively small patient populations and are designed to assess safety and early efficacy. Both drugs will need to complete large Phase 3 trials before they can be considered for FDA approval, a process that typically takes several more years.
If you are currently battling C. diff or recurrent C. diff, please speak with your physician about your treatment options, including if you may be eligible for any active clinical trials. Resources like our Featured Research page and ClinicalTrials.gov can help you and your doctor explore enrollment opportunities.




Leave a Reply